Calcium in daily life
2023-11-07
Calcium in human bones is found in the form of hydroxyapatite. The main component of hydroxyapatite is calcium phosphate, that is, a crystal composed of calcium cations and phosphate anions. The two are so tightly bonded that it's hard to detach them. Although calcium phosphate, like sodium chloride and potassium chloride, are ionic compounds, they differ in the strength of their ionic bonds. According to Coulomb's law, the magnitude of the force acting on a positive and negative charge is directly proportional to the amount of electricity charged and inversely proportional to the square of the distance between the centers of the charge. The calcium ions in calcium phosphate have two positive charges and the phosphate negative ions have three negative charges, which makes the force of attraction between them much greater than sodium chloride or potassium chloride which has only one positive or negative charge. Because of this, tough bones are not easily soluble in water, so if you want to drink bone broth to supplement calcium, not only will it have little effect, but it will consume too much fat.
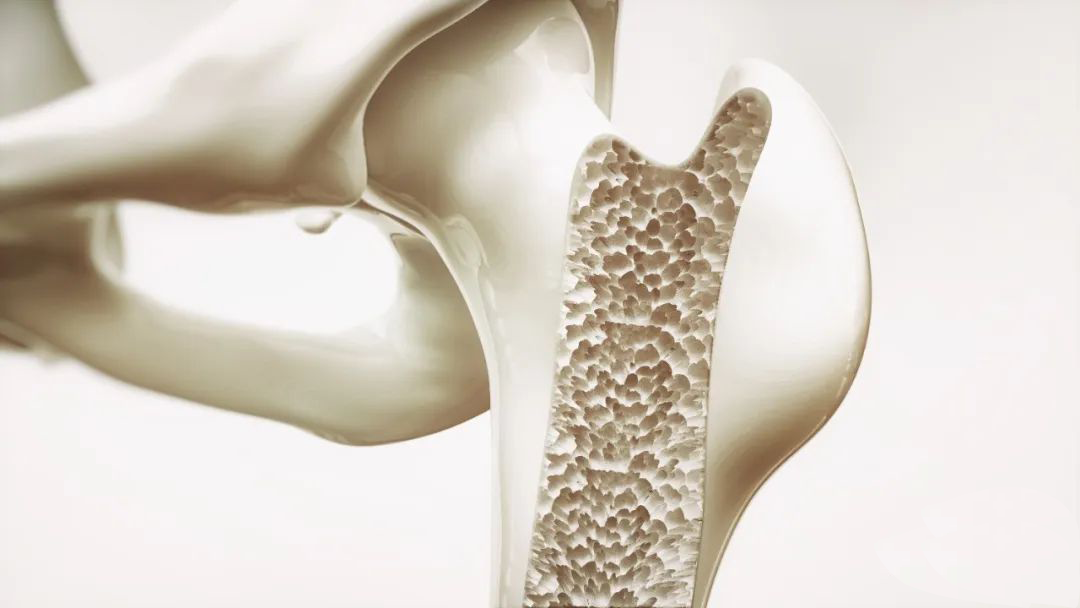
However, when the bones encounter acid, something different. Acids contain hydrogen, and hydrogen ions can get very close to the negatively charged phosphate, so that the gravitational force between the charges increases and forms hydrogen phosphate, dihydrogen phosphate and even phosphoric acid, which makes calcium phosphate soluble. To prevent tooth decay, people need to reduce their intake of acidic foods and foods high in sugar. Under the action of oral lactobacillus, acidic foods can produce more lactic acid, which further demineralizes the teeth and creates cavities. Sugar is the main source of energy in the body and an important risk factor for the development of dental caries. Sucrose is the main cause of tooth decay, followed by glucose, starch, etc. After ingesting too much sugary food, the remaining sugars in the root of the tooth will ferment and become sour at the right temperature, which will then invade the tooth, demineralize it, and cause cavities.
Calcium is the main inorganic component of human bones and teeth, and is necessary for neurotransmission, muscle contraction, blood clotting, hormone release, and milk secretion, among others. Therefore, the normal human body needs to supplement calcium every day (of course, excess calcium will also affect growth and health). So, is it possible to supplement calcium by intaking calcium? The answer is no. Because the absorption rate of calcium by the human body is not high.
In fact, it is very easy to consume calcium, milk, calcium tablets, calcium gluconate, calcium citrate, multivitamin tablets all contain calcium, etc. However, the body's absorption of calcium ions needs the help of vitamin D, and vitamin D in the human body needs to be exposed to sunlight on the human skin before it can be "secondary hydroxylated" to become dihydroxyvitamin D3, thereby promoting the absorption of calcium. Therefore, it is more important to supplement calcium with vitamin D and receive outdoor sunlight at the same time.

In addition to dairy products, calcium-rich foods include green leafy vegetables, legumes, nuts, shellfish, and shellfish, shrimp, crab, etc. Also, don't eat too much salt and oil, as they can also affect calcium absorption.
99% of the calcium in the human body is found in bones and teeth, and only 1% is in the blood, but it is this small amount of blood calcium that plays a very important role. The normal blood calcium concentration in the human body is 2.25~2.75 mmol/L. Low blood calcium is mostly related to hypoparathyroidism, rickets, small intestine malabsorption syndrome, etc., and patients are often accompanied by increased neuromuscular excitability, such as easy agitation, numbness around the mouth and fingers, tetany, muscle tics and other symptoms. High blood calcium is mostly related to hyperparathyroidism, multiple myeloma, kidney cancer, lymphoma, etc., and patients will have symptoms such as weakness, headache, back and limb pain, thirst, and polyuria. Most residents in China generally have insufficient calcium intake, so we need to mix the diet reasonably to improve calcium intake and absorption.
Some green leafy vegetables are rich in vitamins and calcium, but at the same time contain high oxalic acid, which is easy to combine with calcium in the human body to form calcium oxalate, which is not easily soluble in water, resulting in the formation of local stones in the body. Therefore, when eating vegetables with high oxalic acid content (such as spinach, leeks, parsley, green peppers, cabbage, etc.), you should first blanch them in boiling water to remove some of the oxalic acid.
In addition to diet, there are many things in life that are related to calcium. Civil construction, water conservancy, national defense and other projects are inseparable from lime and cement, which are the most commonly used adhesives in the construction field. Quicklime (mainly calcium oxide) is combined with water to form hydrated lime (mainly calcium hydroxide), which then reacts with carbon dioxide in the air to produce hard calcium carbonate. Clay and limestone are important raw materials for firing cement. The main components of cement are tricalcium silicate, dicalcium silicate, tricalcium aluminate and tetracalcium iron aluminate, which will undergo complex hydration and hardening reactions after adding water, and can be well combined with sand, stone and steel to withstand sufficient stress.
In addition, many of the clothes that people wear also have calcium-containing ornaments, such as buttons made of shells or pearls. As for pearl jewelry such as pearl necklaces and brooches, not to mention, their main ingredient is calcium carbonate. It seems that people's clothing, food, housing and transportation are inseparable from calcium.


